| Policy | |
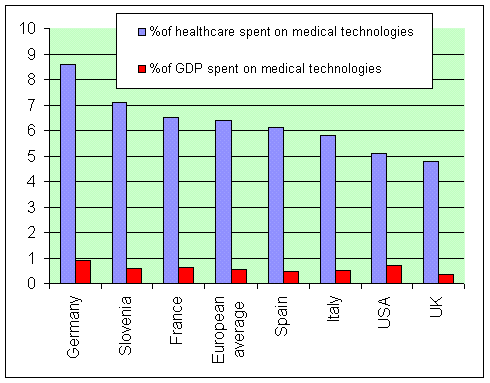
UK government hinders uptake of vital medical technologies3 September 2005 Only a few months after a UK parliamentary report highlighted the need to make better use of medical technologies to improve healthcare, the government is scrapping the committee that assess these technologies for use in the National Health Service. Cuts to the National Institute for Clinical Excellence (NICE) will mean life-saving devices and equipment will be held back for patients in England and Wales, the Association of British Healthcare Industries (ABHI) has warned. The decision to scrap one of the three NICE committees that assess new medical technologies will lead to a massive slowdown in new innovations reaching patients. NICE attributes this decision to £3.5 million cuts in funding. However, the ABHI argues that with the current historic investment in health it makes no sense to cut the gateway to more effective and cost efficient technologies and devices. The ABHI also rejects the Government's argument that the decision can be justified by a lack of new technologies seeking referral to NICE for assessment. The UK government has greatly increased the budget for the adoption of IT, especially in England, through the National Programme for Information Technology (NPfIT). But the UK remains one of the slowest adopters of new medical technologies in the developed world, investing less than the European average in the newest innovative devices — as a recent Health Select Committee report highlighted (1), and as is shown below (data from same report).
Dr Felicity Harvey, Head of Medicines, Pharmacy and Industry Group, Department of Health, told the House of Commons Health Select Committee: "The NHS has not been good at getting new technology in. In its memorandum the Department pointed out that NICE has made a contribution to improving the NHS’s understanding of the clinical efficacy and cost-effectiveness of new medical technologies, but recognised that “more needs to be done to improve implementation of NICE guidance, a point that we have identified, and made recommendations about, in several of our previous inquiries." The government and industry have sought to tackle the problems of uptake of technologies through the Healthcare Industries Task Force (HITF). HITF initiatives aim to speed up development and adoption of new medical technologies within the NHS. The ABHI expressed concern that limiting NICE's assessment ability will potentially limit the impact of HITF and therefore hurt patient care and long-term savings to the NHS. A report published by HITF in 2004 (2) highlighted the benefits and potential savings from medical devices with the example of a device produced by a small British company to analyse the ECG of heart patients:
John Wilkinson, Director General of the ABHI said: "It clearly makes no sense to invest heavily in health but at the same time to massively cut investment in making sure the most effective and cost efficient treatments are used. It is certainly not the case that there is a lack of potentially life saving devices and systems that need evaluation." "We therefore welcome NICE's acknowledgement that cutting its capacity will hamper patient access to new medical innovations in England. We would be more than happy to sit down with them and the Government to address this challenge." Reports 1. House of Commons Health Committee. The Use of New Medical Technologies within the NHS (233K PDF). 2005. 2. Healthcare Industries Task Force. Better health through partnership: a programme for action (534K PDF). 2004. Useful links ABHI: www.abhi.org.uk NICE: www.nice.org.uk |